Published March 2026 | Valen Research | For Educational and Informational Purposes Only
What Is BPC-157?
BPC-157, known as the "Body Protection Compound," is a pentadecapeptide isolated from human gastric juice that has demonstrated pleiotropic effects across various preclinical models examining tissue injury, inflammatory conditions, and central nervous system research. MDPI
It is a synthetic compound consisting of exactly 15 amino acids, making it one of the more compact peptides currently under active scientific investigation.
What Researchers Are Studying
Tissue Repair Mechanisms
Experimental evidence reveals that BPC-157 has been studied in relation to angiogenesis, collagen synthesis, fibroblast activity, and modulation of nitric oxide pathways — areas of investigation that researchers have connected to healing processes in muscle, tendon, ligament, bone, and gastrointestinal tissue. MDPI
A 2025 systematic review found that in preclinical models, BPC-157 improved functional, structural, and biomechanical outcomes across muscle injury, tendon rupture, ligament tear, and bone fracture models.
Inflammatory Pathway Research
Across 36 studies meeting inclusion criteria, BPC-157 consistently demonstrated reduction of pro-inflammatory cytokines while modulating pathways involved in cell growth and angiogenesis. Yourpeptidebrand Researchers have noted that rather than suppressing inflammation entirely — which can interfere with normal healing — the compound appears to support a more organized inflammatory response in preclinical settings.
Gastrointestinal Research
BPC-157 is a pentadecapeptide isolated from human gastric juice that has demonstrated effects in various preclinical models examining inflammatory bowel conditions and mucosal integrity. MDPI This gastric origin has made it a subject of interest for researchers investigating gut-related biological processes.

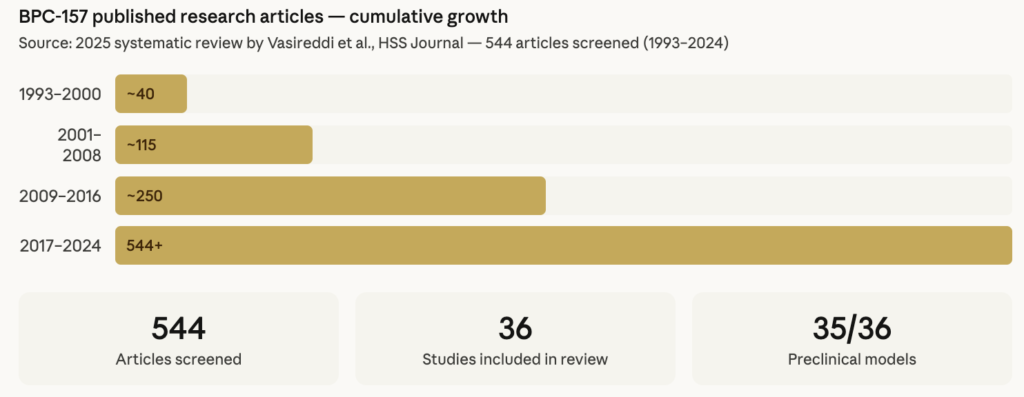
A 2025 systematic review identified 544 articles on BPC-157 published between 1993 and 2024. Of the 36 studies that met inclusion criteria, 35 were preclinical and only one was a clinical study — a ratio that reflects the current state of the research and the significant gap between laboratory findings and human clinical evidence.
This distinction is important context for any researcher engaging with BPC-157 literature.
Most Recent Human Safety Investigation — 2025
The most recent human study, conducted by Lee and Burgess in 2025, was a pilot study involving two healthy adults who received intravenous BPC-157 infusions up to 20mg, with plasma levels returning to baseline within 24 hours and no adverse effects reported. PubMed Central The authors note this represents extremely preliminary data and that large-scale safety investigations have not yet been conducted.
Regulatory Context — February 2026
In February 2026, HHS Secretary Robert F. Kennedy Jr. announced that approximately 14 of 19 Category 2 peptides would be moved back to Category 1, with BPC-157 among the peptides expected to return to legal compounding status through licensed compounding pharmacies with a valid prescription. This reclassification does not make BPC-157 an FDA-approved drug — it means licensed compounding pharmacies may legally prepare it pursuant to a valid prescription from a licensed provider. Thebh
Research Context Disclaimer
All information in this post is drawn from published peer-reviewed scientific literature and is provided for educational purposes only. BPC-157 available through Valen Research is classified strictly as Research Use Only (RUO). It is not intended for human or animal consumption, is not FDA-approved, and is not intended to diagnose, treat, cure, or prevent any disease.
References
Vasireddi N et al. HSS Journal, 2025. PMID: 40756949
Yuan C et al. Int J Mol Sci, 2026. DOI: 10.3390/ijms27062876
Lee E & Burgess K. Alt Ther Health Med, 2025. PMID: 40131143
Rahman et al. J Am Acad Orthop Surg, 2026. DOI: 10.5435/JAAOSGlobal-D-25-00236
For research use only. Not for human or animal consumption.



